How does a water bear survive desiccation?
Loading...
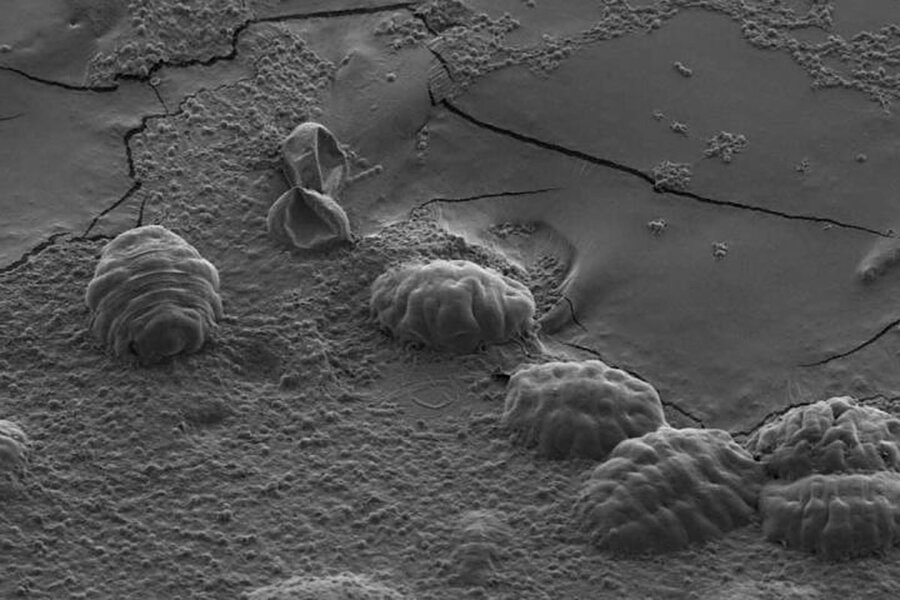
Tardigrades, commonly called water bears, may be too small for the human eye to spot, but that doesn't mean they're delicate. These tiny animals are famous for their ability to survive boiling water, being frozen for years, and even going into space.
They can also survive their cells drying out – an incredible feat, considering all living cells are mostly water. So how do the little animals do it?
Scientists had an idea how tardigrades might survive desiccation, or drying out, because other organisms like yeast and bacteria can also do it. But upon further investigation, researchers found there wasn't enough of the key ingredient – a sugar called trehalose – in the microscopic animals for the suspected process to work.
So a team of researchers decided to look at tardigrades' genes for clues. And now, in a paper published Thursday in the journal Molecular Cell, the scientists report that the little water bears have special genes that encode for disordered proteins that help them survive drying out.
Although this newly described process is protein-based and the other one is sugar-based, the researchers say the mechanism for protecting the tardigrade cells from desiccation is probably about the same.
The idea is that these proteins fill up the cells of the tardigrade and, as it dries out, turn into a glassy substance in a process called vitrification. "What this glass is thought to do is basically trap desiccation-sensitive molecules in this glassy matrix and physically prevent them from breaking like they normally would if they weren't protected," study lead author Thomas Boothby of the University of North Carolina explains in a phone interview with The Christian Science Monitor.
That's how the process is thought to work with the trehalose in yeast and bacteria, too.
"That's probably the most exciting thing," Dr. Boothby says. "There's a good solution, which is vitrification, for surviving desiccation. And nature has basically found two ways to do that. One way uses a sugar, and the other way uses a completely different molecule, a protein. But both kind of go to the same ends."
Traces of that special sugar, trehalose, had been found in tardigrades when some researchers looked, and none when others looked, so scientists were a bit puzzled about whether the sugar helped the microscopic organisms survive drying out.
The other organisms in which trehalose is linked to desiccation survival are flooded with the sugar when they start to dry out, so that about a fifth of their dry body weight is the sugar, Boothby explains. In tardigrades, the most that has been detected has been about 2 percent of the little animals' dry body weight.
Boothby and his colleagues dug into a tardigrade's genome looking for which genes were activated when the animal started to dry out. "What we saw was that the genes that become expressed at the highest levels in the animals when they're drying out belong to a family of genes that are unique to tardigrades," he says.
To test if they had found the right genes, the team reduced the expression of those genes. Sure enough, tardigrades don't survive desiccation very well if the expression of those genes is reduced.
But that wasn't enough for the team. They then tried sticking those same genes in desiccation-tolerant yeast and bacteria. "We found in some cases that expressing a single one of these tardigrade proteins in either yeast or bacteria was able to increase the desiccation tolerance of those systems by up to 2 orders of magnitude," Boothby says. "So that's like 100 times more desiccation tolerant."
The team even tested their findings outside of any organism. They put the proteins encoded by these genes into a test tube along with a desiccation sensitive enzyme, and then dehydrated the whole thing and rehydrated it. And even in a test tube, these proteins wielded their protective power, stabilizing the desiccation-sensitive material during the drying process.
"This is a well done, convincing piece of research that provides new insights on how tardigrades survive [desiccation]," writes John Crowe, a cellular biologist and professor emeritus at the University of California, Davis who was not involved in the research, in an email to the Monitor.
But Dr. Crowe isn't convinced that the disordered proteins are necessarily tardigrade-specific, as Boothby and his colleagues suggest. "It may well be that they will turn up in other anhydrobiotes [desiccation-tolerant organisms], and I rather suspect that they will, or at least that some analog will," Crowe says.
And, he adds, it may not be that an organism can have only one process to survive desiccation. "There may be a continuum from those that use trehalose alone, those that use a combination of trehalose and proteins, and those that use disordered proteins alone," says Crowe, whose own research has used trehalose to increase the shelf life of donated blood platelets from days to years.
There might also be other pieces to that puzzle, too, suggests Peter Alpert of the University of Massachusetts, Amherst who was not involved in the research, in a phone interview with the Monitor. Perhaps the fluidity of cellular membranes may play a role as well, he says.
Ralph Schill of the University of Stuttgart in Germany, who was not involved in this study, agrees that vitrification might not be a sufficient explanation for the desiccation tolerance seen in tardigrades.
Furthermore, Dr. Schill says, "the tardigrade species they used, Hypsibius dujardini, is not a perfect model organism to study desiccation tolerance, because of the very low survival rate after desiccation." Instead, he suggests focusing on the particularly desiccation- and freezing-tolerant tardigrade species, Milnesium tardigradum, that is the subject of his own research.
Still, he says, "tardigrades are really fascinating animals and the new publication is an important piece of the big puzzle of the nature of life."
Studying how organisms can survive drying out is compelling in itself, Dr. Alpert says, but when considering life that can tolerate these extreme conditions, "you wonder, well, could things that don't do it be engineered to do it?"
"People want to engineer crop plants to be desiccation tolerant, for example," says Alpert, whose own research has focused on desiccation tolerance in plants. As drought causes much of the world's famine, engineering plants to survive deep droughts could have a significant impact.