China's lock on market for rare earth elements: Why it matters
Loading...
From Capitol Hill and the Pentagon to high-tech labs and assembly plants, seldom have such obscure names been such a hot topic of discussion. Names such as dysprosium, gadolinium, and praseodymium.
Prase-o-what?
They are mined from Earth's crust, and they are among a unique group of chemical elements vital to making many high- and low-tech products – from iPhones, hybrid cars, and jet-fighter engines to aluminum baseball bats. Such elements also are deemed essential to fielding a new generation of green-energy technologies.
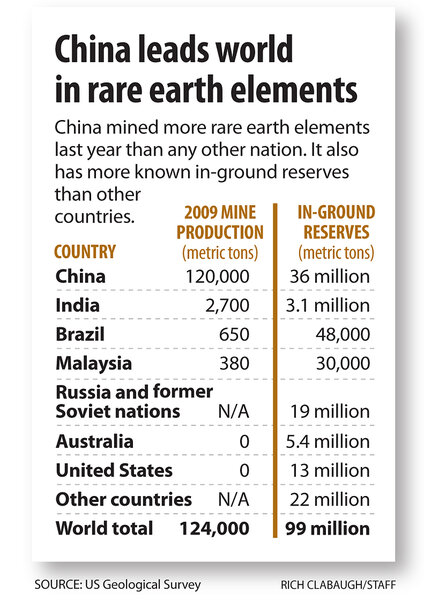
The concern: The US imports almost all of these rare earth elements from China, which currently produces about 97 percent of the world supply. If China were to cut exports for geopolitical leverage, or reduce and tax exports in anticipation of meeting burgeoning internal demand, prices for these elements - and the products they appear in - could jump.
Indeed, over the past 24 hours reports from China have indicated the country could reduce its export quotas by 30 percent next year, following a 72 percent reduction during the second half of this year. Others suggest China might ease up a bit on export limits. And Japan has felt China's rare earth wrath for the past several weeks after China stopped exports of rare earth elements to the island nation over a boundary dispute.
If sustained, such moves could turn some of the most desirable rare earth elements into "unobtainium," at least for several years while mining in other countries ramps up, some experts warn.
What are rare earth elements?
They are a group of 17 elements buried in the middle of the periodic table of elements. The first was discovered in 1787.
Through the early 1940s, they were largely a chemist's curiosity. But then US chemist Frank Spedding figured out how to separate and purify rare earth elements. Twenty years later, researchers began discovering their usefulness.
Despite the name, a throwback to their initial discovery, these elements are as abundant as tin or nickel. But they are tough to mine and extract.
Deposits appear worldwide. The US Geological Survey (USGS) puts total global reserves at 99 million metric tons. China hosts some 36 million metric tons. US reserves are estimated at 13 million metric tons.
What makes these elements special?
They have some handy electrical, optical, and thermal properties, giving them a Midas-like touch when combined with more-common materials.
Europium, for instance, helped drive the shift from black-and-white to color TV by turning the dull reds in early TV screens to reds that popped. Erbium placed at intervals along fiber-optic lines amplifies the light carrying data, allowing it to travel long distances.
Several rare earth elements are used to make smaller, more powerful magnets for cars, computer disc drives, motors, generators – and missile guidance systems. Other materials are used to improve heat resistance in jet-engine turbine blades and provide brighter views in night-vision goggles.
What's all the fuss about?
Four words: single source of supply. It's not a new concern. The issue has been simmering for the past decade. The new wrinkle "is the prospect of an explosion in demand for certain relatively obscure elements for new clean-energy technologies," says Roderick Eggert, a minerals economist at the Colorado School of Mines in Golden.
The Obama administration is pushing development of a new generation of wind turbines, electric or hybrid cars, and electricity-generating solar cells. But so is China, which analysts say is jockeying for position as the world's Wal-Mart for alternative-energy technologies.
Although the US and other countries served as major sources for rare earth minerals for 50 years, China's low labor costs and lax environmental rules allow it to produce both raw and refined minerals at much lower costs than elsewhere.
Undiscovered global deposits are thought to be enough to meet anticipated future demand, according to the USGS. The issue is whether new mines can be on line in time to meet an expected 40,000-ton-per-year shortfall in supply in the next five years.
The situation is not yet a crisis, Dr. Eggert says, but it is cause for concern. Others argue that the situation is more urgent.
Are there viable substitutes for rare earth elements?
They exist for some rare earth elements in some applications. But the wannabes typically don't perform as well, many researchers say.
That leaves several alternatives.
•Establish strategic reserves of the minerals, at least for national security uses. That could help bridge supply interruptions, in the event of mine disasters or if China uses the minerals as political leverage. Japan, for instance, has been laboring for several weeks under a rare-earth-mineral export ban China imposed in the wake of an incident involving a Chinese fishing-boat captain and a Japanese Navy patrol boat. (China denies that the ban was motivated by the incident.)
•Ramp up domestic sources. Bills in Congress would increase efforts to mine rare earth minerals at home, but none has moved out of committee.
•Boost imports from other rare-earth-mineral-exporting nations.
Over the long term, researchers are looking to synthetic substitutes, says Samuel Bader of the materials-science division at the Argonne (Ill.) National Laboratory. Scientists are progressing. Just this year, the American Physical Society awarded its new-materials prize to two researchers for designing and producing a material with unique electronic properties.
To create the material, the scientists described the properties they sought to a program that runs on a supercomputer, and it gave them the design for the material.