Solved? Scientists may have cracked the code of metallic hydrogen
Loading...
With just one proton and one electron, you’d think there isn’t much to learn about the simplest atom in the universe. Yet hydrogen is full of secrets, and a Harvard team says they’ve cracked one more.
Scientists have been chasing hydrogen’s elusive metallic form for over 80 years, foiled by the confoundingly high pressures required to create it. Now, researchers have caught the tell-tale glint of metal in their apparatus, according to a paper published Thursday in Science. Experts consider this novel state of hydrogen a “holy grail” of solid state physics for its potentially world-changing applications, from big bang-for-your-buck rocket fuel to ideal electric wires.
It starts with the universe's most common atom. “We all love hydrogen,” physicist Gilbert Collins of Lawrence Livermore National Laboratory, who was not involved in the research, told Science News in August. “It has the essence of being simple, so that we think we can calculate something and understand it, while at the same time it has such a devious nature that it’s perhaps the least understandable material there is.”
In any environment humans could survive, hydrogen exists as a gas. But just as water can transform into ice at low temperatures, hydrogen gas, like most substances, can become liquid or even solid if you cool it or squeeze it enough. Physicists call these transformations “state changes,” and they go far beyond the solid, liquid, and gas phases many study in school. Even good old water has been shown to have at least a dozen forms beyond the familiar three.
The key to the alchemy of hydrogen is pressure. By definition, metals share electrons that can flow freely throughout the substance, which is why metal spoons get hot while wooden spoons don’t. This property also makes them good at carrying electricity, as well as heat. To get hydrogen to perform this trick, scientists had to squeeze the atoms so hard that electrons got knocked loose from their original atoms, becoming free to move around.
But achieving such high pressures isn’t easy. Early estimates predicted that hydrogen would become metal at 250,000 times the atmospheric pressure of sea level, but as scientists devised ever craftier experiments, the goal kept receding just out of reach.
"There have been dozens of theoretical papers, and they all have different critical pressures for when it becomes metallic," paper co-author Isaac Silvera told Live Science.
It's a question that can only be answered experimentally, and scientists around the world have been racing to create the exotic form of hydrogen in the lab.
“If you look at the literature for the last 30 years,” Eugene Gregoryanz, a physicist at the University of Edinburgh, previously told Science News, “I think every five years there is a claim that we finally metallized hydrogen.” Skepticism surrounding this latest announcement is high, too. At least five experts already told Nature they don’t believe the claim.
But even mistaken claims move the field forward. In the last 40 years, physicists have learned of at least four forms of solid hydrogen, one as recently as 2011, and a 2015 experiment led to a record-breaking new superconductor.
This time, however, Silvera thinks his team has really done it. "No one has ever encountered metallic hydrogen because it's never existed on Earth before," he told Live Science.
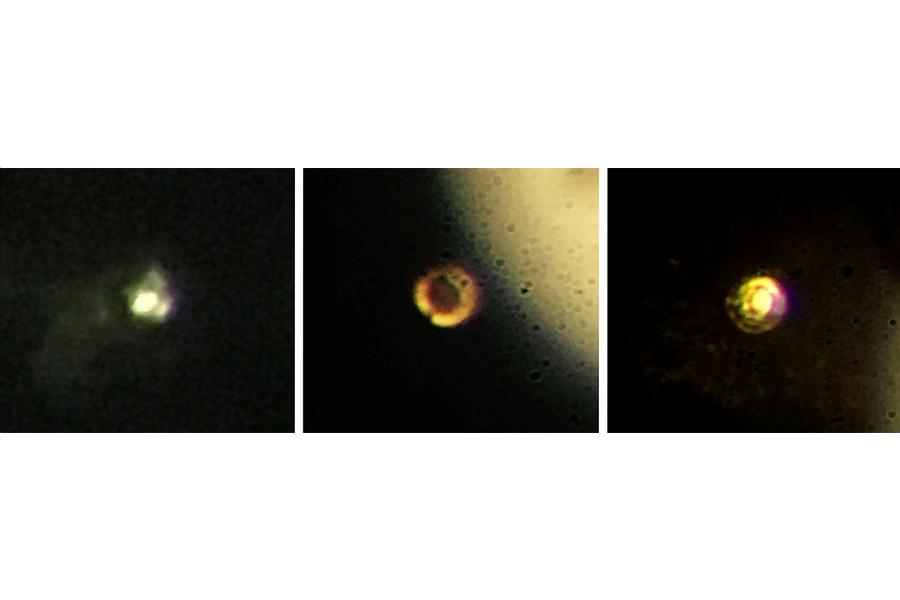
"It was really exciting," he said in a press release. "[Post doctoral fellow] Ranga [Dias] was running the experiment, and we thought we might get there, but when he called me and said, 'The sample is shining,' I went running down there, and it was metallic hydrogen."
The magic number seems to be almost 5 million times atmospheric pressure, 20 times early estimates. Far surpassing the pressure at the center of Earth’s core, you’d have to take a deep dive into Jupiter, which some scientists believe harbors oceans of liquid metallic hydrogen, to experience such a harsh environment.
To achieve such conditions on Earth, Silva’s team took solid hydrogen at minus 452 degrees Fahrenheit (about 20 degrees below its freezing point, and about 8 degrees above absolute zero) and crushed it between two diamond anvils. Keeping even the hardest substance on Earth from shattering at millions of times the atmospheric pressure is devilishly difficult, so the team resorted to a number of tricks, such as applying a special coating to keep the hydrogen from seeping into the diamond, and polishing the surface with a chemical process rather than the typical diamond powder, to avoid leaving atom-sized divots in the surface.
"We designed the system so that all the things that can lead to the breaking of a diamond were not there," Silvera told Live Science.
The substance currently awaits further testing in the diamond vise that created it, but one of the more tantalizing predictions will be tested when the team removes the pressure. Previous work suggests that metallic hydrogen could survive at normal temperatures and pressures, which would have immense consequences for the fields of electronics, magnetics, space propulsion, and more.
"One prediction that's very important is metallic hydrogen is predicted to be meta-stable," Silvera said in a press release. "That means if you take the pressure off, it will stay metallic, similar to the way diamonds form from graphite under intense heat and pressure, but remain a diamond when that pressure and heat is removed."
If true, it means the metal could serve as a superconductor, a material that carries electricity with near-perfect efficiency. Found in a variety of advanced electronics including Magnetic Resonance Imaging (MRI) machines and futuristic Maglev trains, superconductors have been limited in their utility by the requirement that they operate only in very cold conditions.
The current record holder works in environments as warm as -94 degrees Fahrenheit, which is excellent news for residents of Antarctica.
For the rest of us, a room temperature superconductor could mean an end to losing the 5 to 10 percent of energy wasted by resistance in power lines each year. That might sound low, but could be enough to offset all the gasoline the US burns in a year, according to the collaborative journalism initiative Inside Energy.
Another potential application of the new material is rocket fuel, because its concentrated energy could pack more than three times the punch of standard propellants.
Metallic hydrogen “would easily allow you to explore the outer planets,” Silvera said. “We would be able to put rockets into orbit with only one stage, versus two, and could send up larger payloads, so it could be very important.”
But for now, it’s back to the lab to test the sample’s stability and conductivity. After more than four decades of work on metallic hydrogen, Silva said he’s thrilled to see the results.