Did our fingers evolve from fish fins?
Loading...
We were all fish once – well, our ancestors were. But how did ocean swimmers evolve to scuttle around on land?
It would have taken a certain dexterity that fins wouldn't have allowed, but fingers and fins seem like such different structures. Scientists have long puzzled over this particular element of the evolutionary leap to land, but now they think they have an idea of how it happened: the same cells that play a role in developing fin bones also contribute to the formation of digit bones.
And this discovery could change how scientists see this major evolutionary transition.
It seems "so utterly improbable and impossible" that fins somehow evolved into limbs, study senior author Neil Shubin, a paleontologist and evolutionary biologist at the University of Chicago, tells The Christian Science Monitor. But as it turns out, "these aren't as giant steps as you'd think by looking just at the endpoints."
The new research is detailed in a paper published Wednesday in the journal Nature.
Human limbs, say biologists, evolved from the fins of their fish ancestors some time between 385 and 360 million years ago, scientists know. But what they don't know is how exactly that happened, or how fingers figured into the fishy puzzle.
You might imagine that the bones in a fish fin (called fin rays) evolved into fingers and toes, and that the bones connecting the fin rays to the rest of a fish's skeleton became wrists and ankles. But fin rays are actually a different kind of bone. They're dermal bones, while human digits are cartilaginous bones: structures fundamentally different enough that they formed through a completely different mechanism.
So if fin rays are not "the ancestors of digits," Clifford Tabin, an evolutionary geneticist at Harvard University who was not part of this study, writes in an email to the Monitor, "then where did digits come from?"
"One model would be that the hand is a completely new structure ('neomorphic') in tetrapods. The other option seemed to be that some of the bones at the base of the fin (the 'posterior radials') were expanded and elaborated to make the digits," he writes. "Either way, the suspicion was that there was an entirely new developmental program developed to make them."
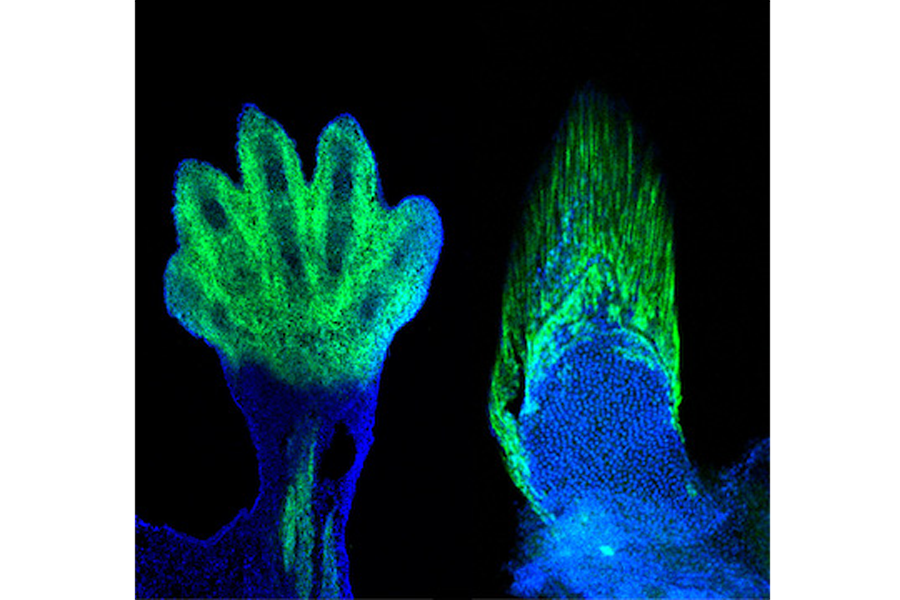
So when Andrew Gehrke, a graduate student in Dr. Shubin's lab, handed Shubin a picture showing where the cells associated with wrist and digit development in four-legged animals (tetrapods) end up in a developing zebrafish embryo, Shubin's jaw dropped.
"It was totally against my expectations," he says. If digit development and fin development were truly distinct processes, "the markers of the wrists and digits should end up in the cartilaginous bones, which sit at the base of the fin, not in the fin rays."
But there those cells were, in the fin rays.
Meanwhile, Tetsuya Nakamura, a postdoctoral researcher in Shubin's lab, was using the newly developed CRISPR/Cas9 gene-editing technique to scramble hox genes, which are associated with limb development in tetrapods, from zebrafish.
It turns out that fin rays, like the toes of mice, are diminished when these genes are edited out.
But that's not all. The cartilaginous bone at the base of the fin had also increased, suggesting that the processes are actually linked – matching what Shubin and colleagues have seen in Tiktaalik roseae, a fossil that is thought to sit on the cusp of the water-to-land transition.
What's happening, he suggests, is that a common population of cells behaves differently in different environments within an animal's body. By changing the hox gene activity, the cells don't migrate out to the tip of the fin to develop the long fin rays found in normal zebrafish. Instead, those cells cluster at the base of the fin to develop more cartilaginous bones in the mutants instead.
This would mean that in fact digit development isn't an entirely separate process from fin reduction as previously thought.
This is still just a hypothesis, Shubin says. "It's supported by a lot of data, but to really nail it, I think we need to watch those cells in the mutants a little bit better."
It will be intriguing to see whether or not these are exactly the same cells forming the extra cartilaginous bones that would usually form a fin ray, says Martin Cohn, an evolutionary biologist at the University of Florida who was not part of the study. If they are the same cells, as Shubin and his team suggest, this could have broader evolutionary implications.
"Although the structures themselves might not be homologous," Dr. Cohn tells the Monitor, "there could be homology at the level of cell-type identity."
In other words, even if two anatomical structures appear to be completely different – like, say, fin rays and fingers – "the progenitor cells and the genetic circuitry responsible for building those structures might have been present in the common ancestor," Cohn says. This sort of cellular homology could help explain other evolutionary conundrums as well, he adds.
As for the proto-tetrapods swimming around in the Devonian period, as creatures began the journey from water to land, all that it would have taken would have been a switch to signal this circuitry to make more limb-like skeletal structures instead of fin rays, according to Dr. Tabin. The new paper is "an unexpected finding that really adds a new level of clarity to the fin-to-limb transition," he says.