Scientists might have built the world's smallest 'gears'
Loading...
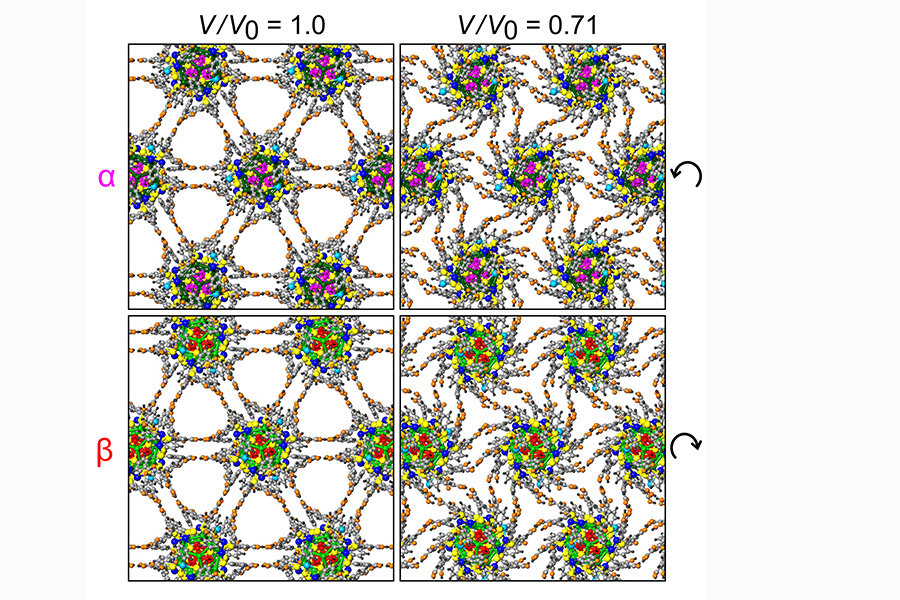
Using silver nanoparticles, scientists have constructed a molecular "superlattice" with an extraordinary property: when subjected to pressure, its tiny parts can rotate in unison.
And this special property could help develop materials that are less susceptible to any damage, says Terry Bigioni, an associate professor from the University of Toledo who was involved with the study.
The scientists carrying out the research, which was supported by the Air Force Office of Scientific Research and the Office of Basic Energy Sciences in the Department of Energy, crystallized nanoparticles, resulting in the formation of a specific pattern in which molecules of nanoparticles occupied the "lattice" points within the crystal.
“It’s not the individual atoms that form the superlattice,” Landman said in a press release. “You actually make the larger structure from clusters that are already crystallized. You can make an ordered array from those," said Uzi Landman, a Regents’ and F.E. Callaway professor in the School of Physics at the Georgia Institute of Technology
Each nanoparticle molecule – connected to each other with hydrogen bonds – consists of a small metal core surrounded by organic molecules.
“As we squeeze on this material, it gets softer and softer and suddenly experiences a dramatic change,” said Dr. Landman. “When we look at the orientation of the microscopic structure of the crystal in the region of this transition, we see that something very unusual happens. The structures start to rotate with respect to one another, creating a molecular machine with some of the smallest moving elements ever observed.”
The rotation of these "gears" could be as much as 23 degrees. The movement is facilitated by hydrogen bonds that basically serve as the "hinges," researchers say.
When the pressure stops, the material goes back to their original state, says Dr. Bigioni.
Unlike most materials that bulge out when hit by a force become denser at the point of impact, Bigioni says, suggesting that they could be of use in projectiles or body armor.
“We make the small particles, and they are different because small is different,” said Landman. “When you put them together, having more of them is different because that allows them to behave collectively, and that collective activity makes the difference.”
The study was published in a paper titled "Hydrogen-bonded structure and mechanical chiral response of a silver nano particle supperlattice" on April 6 in the journal Nature Materials.