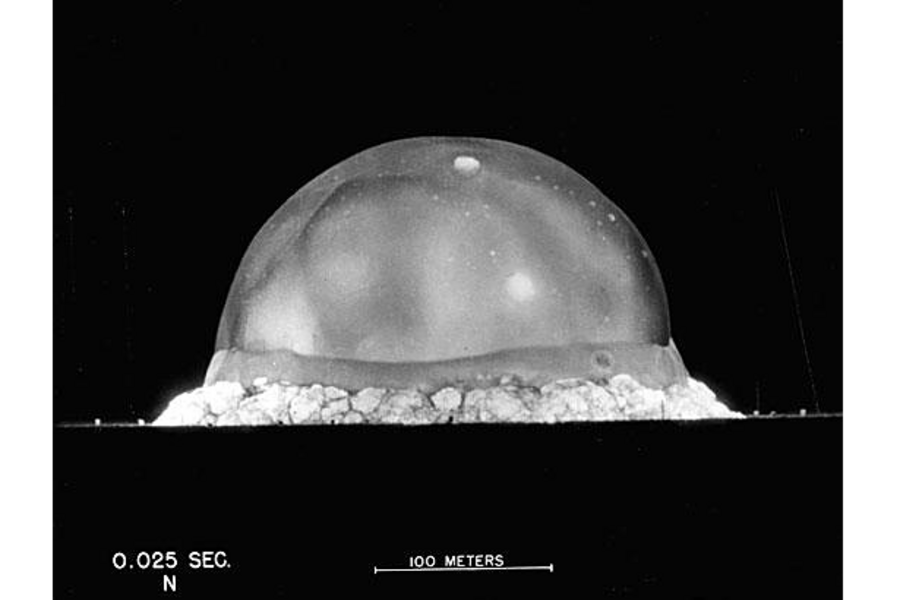
What could a nuclear bomb test site and the moon have in common?
Loading...
How did the moon form? Scientists base their models largely on data from moon rocks and meteorites. But those just provide momentary snapshots, not geological processes in action. To test their models, scientists need to figure out just how, and under what conditions, those rocks may have formed. And one team may have just found a new way of thinking about lunar formation.
Moon-building happens on a massive scale, so "we were looking for an analogue that was large-scale enough ... to replicate what we thought was going on during the early process of planet formation," James Day, a geochemist at the University of California San Diego, explains in a phone interview with The Christian Science Monitor.
Fortunately for Dr. Day and his colleagues, a real-life massive, super hot explosion took place just a few decades ago: the detonation of a nuclear bomb.
And it, too, altered the chemistry of rocks. When the dust settled after a plutonium bomb was tested for the first time near Alamogordo, N.M., in July 1945, some of the red soil had become a light green glass. The glass was dubbed trinitite after the test site, called Trinity.
"We can use the Trinity glasses – that came from this very profound experiment that's had a huge effect on human history – to scientific benefit," Day says.
By studying the trinitite, Day hoped to glean insight into how the material that formed the moon might have changed during, if it formed according to the canonical model of lunar formation.
In that classic "great impact" model, a Mars-sized object slammed into the early Earth, vaporizing some material and blasting other rocks from Earth's surface into a disk in Earth's orbit that then accreted to form the moon.
What does that have to do with a nuclear bomb test?
The impact from the Mars-sized body was probably violent enough to deplete some of the volatile elements in the material that eventually formed the moon, explains Day. Similarly, he thought, the nuclear explosion might have made the trinitite depleted in volatiles.
The researchers focused in on the volatile element zinc. The idea was that the hot, high-pressure conditions of the nuclear explosion would mimic the conditions thought to occur in the great impact model and cause evaporative fractionation. In other words, the lighter isotope of zinc would have been more likely to evaporate in the explosion than the heavier isotope.
Similar fractionation of zinc has been identified in some moon rocks, called mare basalts.
"Our expectation was that when we measured these trinitites, these glasses that formed in the nuclear detonation, that we were either going to see nothing … that in fact, our hypothesis that there was volatile loss during this event was wrong," Day says, "or that we would approach the theoretical values that have been calculated" for the giant impact model, which suggested more loss than the moon rocks displayed.
But, he says, it was "startling" how close the zinc composition of the glasses looked to the lunar rocks.
Day and his colleagues report their findings in a paper published Wednesday in the journal Science Advances.
Does this really tell us anything about the moon?
James Van Orman, a geochemist at Case Western Reserve University who was not part of Day's team studying the trinitite, says it's a "creative and unique" idea to use the products of a nuclear test to better understand how such conditions alter a rock's chemical make-up.
"There is no question that this constraint on Zn isotopic fractionation during evaporation will be valuable in modeling volatile loss from the Moon, and comparing the models to data from lunar samples," Dr. Van Orman writes in an email to the Monitor.
But to draw conclusions about the moon's formation from this one study may be a bit of a leap, he says.
Day agrees that his research can't really say much about the mechanism of how the moon may have formed. "All it tells us is that the conditions that we would expect, of high temperatures, very Hadean-like conditions," he says, "were occurring at the time that the moon formed."
But Van Orman says it might not even address the event that formed the moon.
"This study shows clearly that evaporative loss of Zn can explain the heavy zinc isotopic composition of the mare basalts," he explains. But that doesn't mean it's the only way to explain that composition.
The mare basalts are volcanic rocks that cooled from lavas that may had time to degas before they hardened, he says.
"I'm not convinced that the evaporative fractionation was associated with lunar formation, rather than later magmatic degassing," Van Orman says.
"You don't have to do it during the giant impact," agrees David Stevenson, a planetary scientist at the California Institute of Technology who also was not involved in the study.
"There is a big difference between identifying the results of a process and directly applying the specifics of what happened in the Trinity test with what happens in planet formation," Dr. Stevenson writes in an email to the Monitor. "So I doubt that science by analogy (which is what they're doing) is very useful, but I seen value in their work nonetheless." This new study, he says, "quantifies a process that happens when you severely heat a rock (more precisely a droplet of magma). That's useful."
Jay Melosh, a geophysicist at Purdue University, suggests that older crustal moon rocks, perhaps from the lunar highlands, may have been a better choice than the mare basalts to compare with the trinitite.
Building a moon model isn't always easy
Applying this study to questions about the moon's formation may actually complicate the picture, Dr. Melosh says.
Scientists thought they had it all figured out decades ago, he explains in a phone interview with the Monitor.
But when researchers got better at measuring isotopic differences, they realized the canonical model had a big problem.
Melosh refers to this puzzle as "the isotopic conundrum." In the classic model, researchers would expect to see these isotopic fractionations across other volatile elements in moon rocks, like potassium. "In fact," he says, "we don't."
[Editor's note: Following the publication of this article, Day pointed out that a paper published in Nature in October 2016 has found evidence of fractionation of potassium isotopes in lunar rock.]
Since this isotopic puzzle has arisen, many scientists have tried to tweak the giant impact model to resolve it. Some have proposed that the impact was particularly violent, while others have thought more "out-of-the-box" and suggested that the moon may actually be made up of many mini-moons.
The fact that zinc is isotopically fractionated in the moon rocks but potassium is not "adds to the many mysteries about the moon," Melosh says. "The moon certainly is giving us a lot of puzzles."