Contact lens recall: dramatic expansion for CooperVision
Loading...
In a dramatic expansion of its August contact lens recall, CooperVision has announced that it is recalling 6.6 million units of another line of contact lenses for the same problem.
The problem lenses have been linked to hazy vision and eye discomfort and sometimes severe pain.
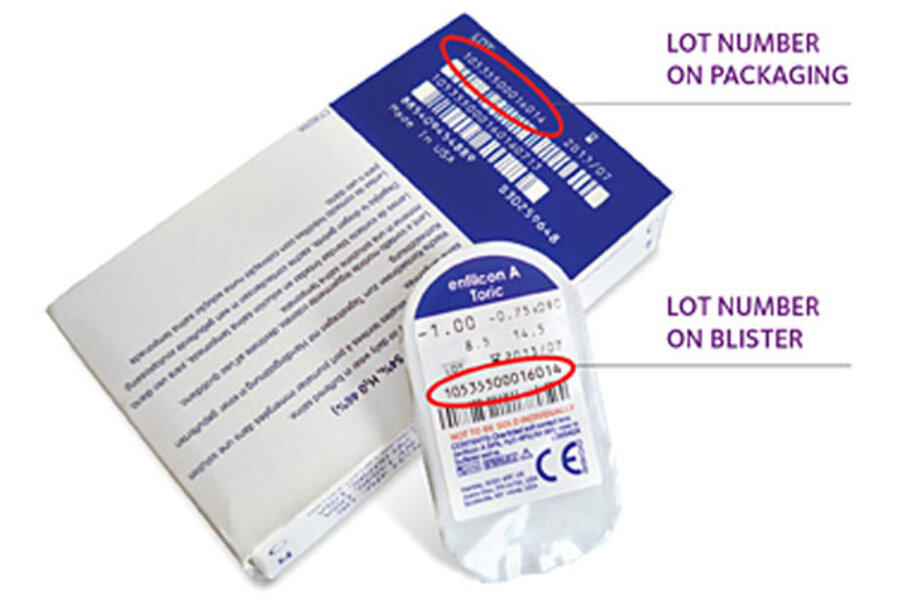
The new recall involves Avaira Sphere lenses. CooperVision is urging customers who have them to check their lenses’ lot number, which can be found on the back of the packaging by the bar code as well as on the individual blister packs, which is just above the bar code. (See the photo above for exact locations.)
To see if their lenses are part of the recall, customers should either call the company toll-free at 855-526-6737 or punch the lot number into the special recall page on the CooperVision website.
If your lenses are on the list, the company says you should stop using them immediately and return them to the eye-care specialist or retail store where you bought them. You will receive free replacement lenses.
The company is not publicly releasing which lot numbers are involved, but it has given them to the eye-care specialists who prescribe the lenses and is urging them to contact their customers directly.
Of the 6.6 million Avaira Sphere lenses recalled, 4.9 million had already been shipped to customers globally. It appears that somewhat less than a quarter of the company’s Avaira Sphere output is affected by the recall.
The problem originally cropped up with CooperVision’s Avaira Toric lenses earlier this year, with customers complaining of hazy vision, discomfort, and more severe problems. That prompted the August recall of 600,000 lenses in the United States.
The company traced the problem to an excess of silicone oil residue in the manufacturing process. So it revamped its manufacturing and quality control.
“We've now put into place a quality process that's much tighter,” says James Gardner, the company’s vice president of marketing for the Americas.
It was this upgraded quality process – and not customer complaints – that caught the excess residue on the Avaira Sphere lenses and prompted this month’s expanded recall, he adds. “We believe that it's the right step to take…. The health and safety of our customers are our top priority.”
CooperVision is a subsidiary of the Cooper Companies, a global medical device company based in Pleasanton, Calif.