Lab that made tainted steroid shots to get 'CSI' treatment
Loading...
Investigators are sifting through records and testing a Massachusetts lab to determine how a fungus associated with a form of meningitis worked its way into products that should have been sterile.
"It's really 'CSI' stuff," says Eric Kastango, president and owner of ClincalIQ, a New Jersey-based consulting firm that aims to help the industry meet safety standards. "The microoganism in the vials is the same microoganism in the patients. The question is: How did it get in the vials in the pharmacy?"
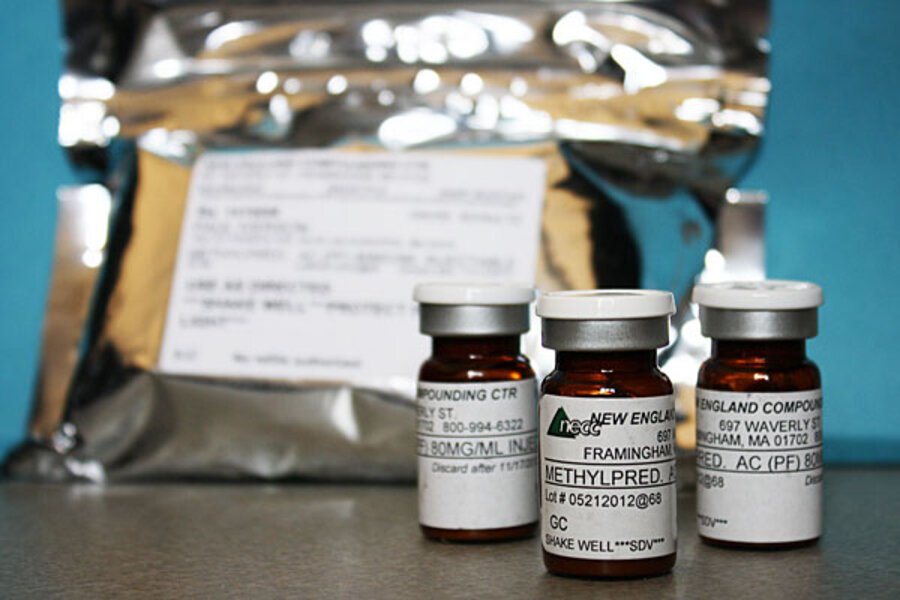
The incident involves the New England Compounding Center (NECC) in Framingham, Mass., which is no stranger to regulator scrutiny. Records released Monday by the Massachusetts Department of Public Health indicate that the company has been the subject of several complaints and inspections since 1999.
In the current case, large shipments of injectable steroid formulated by NECC have sickened 308 people in 17 states and killed at least 23, according to figures updated on Tuesday by the US Centers for Disease Control in Atlanta. Some 14,000 patients in all may have received contaminated injections.
Compounding pharmacies typically produce limited quantities of medicines whose formulation or dose is tailored to patients – pets as well as people – whose doctors deem as inappropriate treatment standard doses of mass-produced medicine. In addition, hospital pharmacies can tap advanced compounding pharmacies for some of the sterile medicines the hospitals use.
The US hosts more than 7,500 pharmacies that provide advanced compounding services, according to the International Academy of Compounding Pharmacies. Of those, some 3,000 produce sterile compounds.
Contamination incidents involving sterile compounds from compounding pharmacies are rare, but they do happen. Between 1990 and early 2012, at least 21 incidents involving contaminated sterile medicines from compounding pharmacies have occurred, killing 25 patients and leaving more than 120 others with serious side effects, according to a list compiled for the Institute of Safe Medication Practices, based in Horhsam, Pa.
In some ways, the potential for at least tiny amounts of contamination comes with any compounding, acknowledges Kara Weatherman, a pharmacist and clinical assistant professor of pharmacy practice at Purdue University in West Lafayatte, Ind.
"You try to avoid the potential for contamination," she says. "But unless you're working in an environment where there are no particles, there's always a chance that something is going to end up in your product."
In the case of microorganisms such as the fungus in the NECC products, the longer a vial sits around waiting to be shipped or used, the longer the microorganisms have to expand their colony. Testing a product before it leaves the pharmacy to ensure it's sterile can in some cases take up to 14 days – which can be impractical if the shipment is urgent.
"In those cases you just say: 'OK, we're only going to use this drug for five days.' You limit the shelf life," she says, to prevent any microbial contaminants from going into population overdrive.
How materials were stored and how long were they in storage are two issues that investigators look into, says Edmund Elder Jr., director of the Zeeh Pharmaceutical Experiment Station at the University of Wisconsin in Madison.
According to Food and Drug Administration records, one of the agency's concerns about NECC in 2006 was that it repackaged sterile, injectable drugs – a process that exposes the sterile drug to contamination as vials are opened and the contents are transferred to syringes. The FDA warning letter also raised related storage issues.
Beyond process issues, the company also has come under scrutiny for the large amounts of drugs it produces.
"We're finding out that volume out of this pharmacy was astronomical," says Mr. Kastango says. The numbers suggest that its operation had crossed the divide between compounding to match prescriptions and out-and-out manufacturing, he adds.
The fungus involved in the NECC case can crop up in homes, soils, water supplies, or the air, says Dr. Elder, who has participated in investigations of previous incidents at compounding pharmacies. It can enter a lab on shoes, on the lab coat of someone outside taking a break and not changing into clean lab garb after returning, or though air ducts, adds Mr. Kastango, pointing to past examples.
Investigators typically aim to pinpoint the pathway by which a contaminant entered the product. But they also eye a pharmacy's environmental-monitoring effort, which is supposed to check for the presence of potential contaminant organisms within the facility. Such efforts are included in a detailed set of international standards, which is the foundation for federal regulations as well as regulations in many states. The standards also call for periodic checks of employees – swab-testing hands and lab suits, for instance.
Inside a facility, some small amount of contamination may be present, specialists say. The key is to make sure minimal contamination remains minimal.
Reflecting on incidents he's helped investigate, Elder draws an analogy to a catastrophic airliner crash: "You can have single things go wrong in any flight, and the plane still lands in the end. But if you have multiple things go wrong, and the pilots involved don't react appropriately, we see a catastrophic event."
"It's not necessarily one thing that went wrong, but a number of different things that went wrong that sometimes have built up over time," he adds."Someone took a shortcut, nothing happened, so that becomes their normal way of operating."